��������������Ӧ����ѧ�о�
��? PB08206281
�й���ѧ����������빤��ϵ
230026����կ·96�ţ��Ϸ�
ժҪ��ѧ����ѧ���о���������ѧ����Ҫ��ɲ��֡�ֱ��ͨ����ѧ�������ⶨ��Ӧ�Ķ���ѧ�������������Խ��еģ���ʵ����ò����絼�ʵķ����������ѧ��Ӧ�����ʳ����ͻ�ܣ����ž�ȷ�Ƚϸߣ�ʵ������ԱȽϺõ��ص㡣
�ؼ���������Ӧ �絼�� ��������˹��ʽ
Study of Kinetics of Ethyl Acetates�� Saponification
Wu Kun
Department of material science and technology��University of Science And Technology China
230026��#96��JinzhaiRoad��Hefei
ABSTRACTAn important part in Physical Chemistry is Kinetics in reactions. Usually, that��s an impossible job to determination Kinetics Constant by chemical analysis ways. We Measured
The Conductivity to know the K an the E, which is an Simpler and Accurate way.
KEY WORDSSaponification Conductivity Arrhenius
1ǰ��
������Ӧ(Saponification)�Ǽ�(ͨ��Ϊǿ����µ�����ˮ�⣬�����������������Σ���ָ��֬��ˮ�⡣����Ľ���������Ӧ��������֬���������ƻ��������ػ�ϣ��õ���֬�������/���κ��͵ķ�Ӧ�������Ӧ��������������е�һ������˶����������Ļ�ѧ��Ӧ������1823�걻������ѧ��Eug��ne Chevreul���֡�������Ӧ����������֬���������Ʒ�Ӧ�⣬������֬��Ũ��ˮ�ķ�Ӧ��
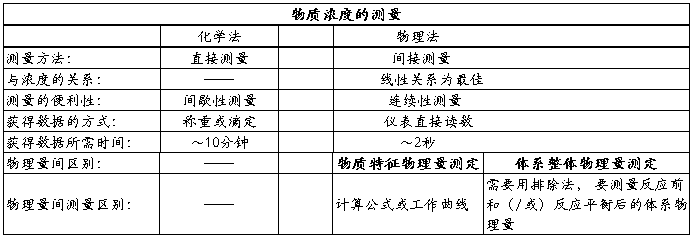
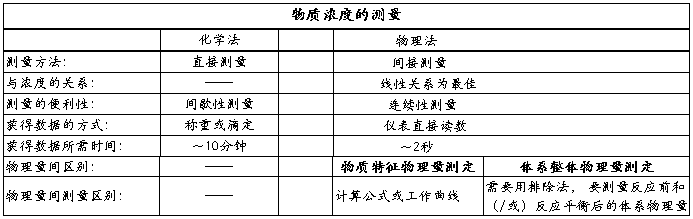
��ʵ�����л�ѧ��Ӧ����ѧʵ��Ĺؼ���ȷ���Ũ����ʱ��仯�Ĺ�ϵ��һ�������ַ�����
1��ѧ���������ŵ㣺ԭ��ֱ�ӣ��������ˡ�ȱ�㣺���������ӳ�ЧӦ������ȷ�Խ��ͣ���ʵ����Ʒ���ƻ������ıȽ�������
2�絼�����ŵ㣺�ٶȿ죬����ȷ��ȱ�㣺��Ҫר����������ʵ��ȷ��������ʵ����������ƱȽϴ���ֻ�ܲⶨ�����Һ�塣
���������ַ����������±����˲���
��ʵ��ʹ���˵絼����
����������������Ӧ��Ϊ1:1�ı�����ϵ�ͱȽ����õ��ķ�Ӧԭ���뷴Ӧ���������Ծ����������о����ڱ�ʵ���У����ڷ�Ӧ���ڽ�ϡ��ˮ��Һ�н��еģ����ǿ��Լٶ�CH3COONaȫ�����룬��Ӧǰ����Һ��������Ŀ������������ɲ��䣬������CH3COO����Ǩ���ʱ�OH����Ǩ����С�����ŷ�Ӧ�Ľ��У�OH�����ϼ��٣�CH3COO����Ũ�Ȳ������ӣ�����ϵ�絼��ֵ����½�����һ����Χ�ڣ�������Ϊ��ϵ�ĵ絼�ʵļ�������CH3COO����Ũ��x�����������ȡ���ˣ��ڱ�ʵ��ʹ�õ絼���ǿ��еġ�
��������������Ӧ����ʽΪ��
? CH3COOC2H5��Na����OH�� ?? CH3COO����Na����C2H5OH
������������������Ӧ��ԭ���������ɹ�ʽ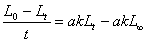 ���k��
���k��
�����ݰ�������˹��ʽ�����Լ��㷴Ӧ�Ļ��
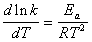
2ʵ�鲿��
��1��ʵ������

��2��ʵ�鷽��
1�����²�ʹ�������30�档
2��NaOH��Һ�����ƣ�
��һ��С�ձ�����������ŨNaOH��Һ�����ش�Ĺ��ƿװ��ܶ�ĵ�����ˮ�����ӽ���ˮ��������ѡ��ʵ�������IJ����缫����ˮ�У���Ž��������£���μ���ŨŨNaOH��Һ��L=1300��1400��S/cm��
3��NaOH��Һ�ĵζ���
�����ƺõ�NaOH��Һ���˹��ֶ��ζ��ܺͷ�ָ̪ʾ���������½���Ũ�Ȳⶨ���ظ��������ϣ�ȡƽ��ֵ��
4��L �IJⶨ���� 30.00�棩
�IJⶨ���� 30.00�棩
ȡ100ml�����ҵζ��õ�NaOH��Һ���ں��¼���Ӧ���У�����ϴ��������ˮ�IJ����缫������10���ӣ��ü������������������������ȡ����
5��L �IJⶨ����30.00�棩
�IJⶨ����30.00�棩
���L �IJⶨ��ʹ��С��������Һ����ȡ��������������������������ڲ����ף�����������ȫ��������Һ�У���Ҫ�����ڲ������ڱ��ϣ�����Ũ�Ȳ����ŵ�һ��ʱ�������ʱ������ƽ�ȱ仯����READ��������ȡ�����ڵ絼����һ���������ں�40min�����������Զ�ֹͣ������
�IJⶨ��ʹ��С��������Һ����ȡ��������������������������ڲ����ף�����������ȫ��������Һ�У���Ҫ�����ڲ������ڱ��ϣ�����Ũ�Ȳ����ŵ�һ��ʱ�������ʱ������ƽ�ȱ仯����READ��������ȡ�����ڵ絼����һ���������ں�40min�����������Զ�ֹͣ������
6��������3��4��5��35.00���½��в�����
��3��ʵ�����ݼ�¼
1�������ȡ�����������Ѿ��ϴ���FTP�ļ��С�
2������������
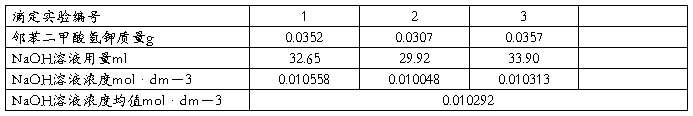
3����õļ���������������Ϊ0.05ml��
��4��ʵ�����ݴ���
�й�ʽ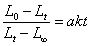 ֪��Ҫ�ó�k����Ҫ����
֪��Ҫ�ó�k����Ҫ����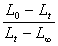 ��t��ͼ��
��t��ͼ��
��30�棬L0�ȶ���1303��S/cm�����ɶ϶�L0Ϊ 1303��S/cm��
��35�棬L0�ȶ���1292��S/cm�����ɶ϶�L0Ϊ 1292��S/cm��
ÿ���Ӷ���12����������40���ӣ������480�����ݣ��������ݱȽ϶࣬���Ե�һ�г�������д������˱���һ���ϴ���
30���϶����ݴ���.xlsx
35���϶����ݴ���.xlsx
Ȼ����origin������ֵ��ʱ������ߡ�
�������������Ϊ
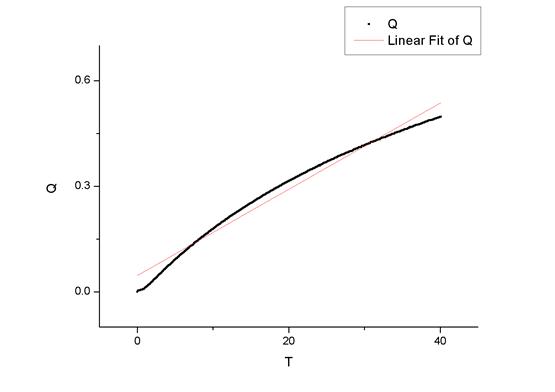
ͼһ��30���϶��������
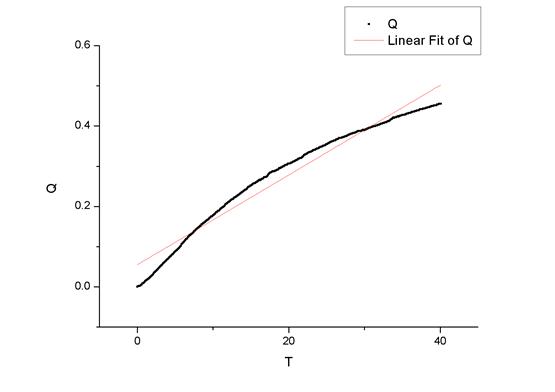
ͼ����35���϶��������
��origin������������ߵ�б��Ϊ

���Է�Ӧ���ʳ���K1=0.9496 dm3��mol-1��min-1��K2=1.0845 dm3��mol-1��min-1��
�����
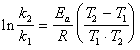
Ln0.911*155180=E
E=-14464J/mol
��5��ʵ��������
1���������ó�����Ӧ�Ļ������Ϊ28286J/mol����ʵ��ֵ���Ƚϴ��������������Ҫԭ���У�
���ȣ������ڸ���35��ʱ�����ʳ�����������30��ʱ����������������Ҫԭ��Ҳ�Dz����������ء�����������������Ӧ�ڱȽϸߵ��¶���Ӧ�÷�Ӧ�ıȽϿ졣Ȼ���۲����ݣ����ָ����µ��������Ǵӷ�Ӧһ��ʼ�����ֳ����ģ�һֱ�������Ա���������ƣ��ɼ�Ӧ���Ǹ���ʵ�����ݵ�żȻ�����ɵġ�
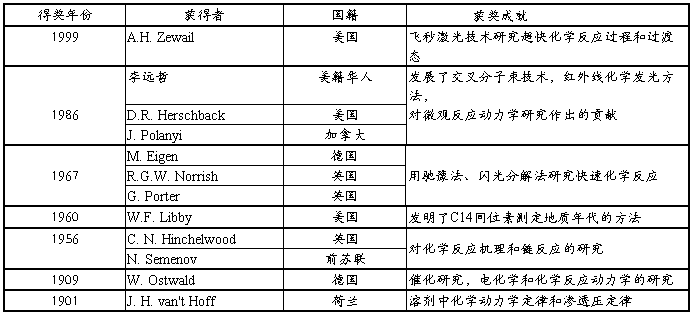
��Σ���˵K1 �ٴΣ�����10���������Ժ�Ӧ��ϵ���¶Ȳ�����������35�ȣ�������34�����ң�Ҳ����˵������ϵ���²�ȼ����е�С�� ��絼������Ҫ��ϴ�����Dz����ֲ��ܲ��ã���Ȼ������������ˮ��������Ⱦ���һ��Ӱ����ʵ������ȷ�ԡ� 2����ͼ�п��Կ�������ֵ��ʱ�����������������ƣ�˵������ʱ������ƣ���Ӧ���г̶���ߣ���Ӧ������еĶ�������������ǰ�����������͡� 3����Ӧ�IJ�ȷ����ʮ�ֶࡣ�����ڼ�������������̫�٣������������������ӷ����ʣ����ʵ�ʼ�������Ƚϲ�ȷ�� 4�����⣬������û�м�������������ʱ����Һ�ĵ絼����Ȼ�Ƚ��ȶ������Գ��ֳ��½������ƣ�������������Ũ�Ⱥܵͣ������ܳ��ֵ��벻���ף���������Ҫ����Ϊ����������Һ�����ڽ����²�����һ������ʧ�� 4ʵ����� ͨ����ʵ�飬���Կ�����ѧ��Ӧ����ѧ�ص��ڱ���Ӧ�����ֵ�ʮ�����ԣ�����С��Ӱ�����ض���Է�Ӧ��ʵ����������ϴ��Ӱ�졣�ڲ����²��С�ķ�Χ�ڣ��Ƚ�����ȷ�����������Ӧ�ķ�Ӧ��ܡ������¶�����20���������Ż��и�Ϊ����Ľ���� 5�ο����� 1����������ѧ���²ᣬ���İ� ���ײʣ�����ϼ��Ҧ����࣬�ߵȽ��������磬1990 2����������ѧ��(��)V.����µ��� �ߵȽ��������磬 1983��7�� 3����չ��21���͵Ļ�ѧ�������ɵ����࣬��ѧ��ҵ�����磬20##��5�� ʵ��ʮ ��������������Ӧ����ѧ�о� һ��ʵ��Ŀ����Ҫ�� ? 1�����ջ�ѧ����ѧ��ijЩ��� ? 2���ⶨ��������������Ӧ���ٶȳ����� 3�� ��Ϥ�絼���ǵ�ʹ�÷����� ����ԤϰҪ�� 1�� �˽�絼���ⶨ��ѧ��Ӧ�ٶȳ�����ԭ���� 2�� �˽�DDS-�͵絼���ǵ�ʹ�÷����� ����ʵ��ԭ���� ? ��������������Ӧ����ʽΪ�� ? CH3COOC2H5��Na����OH��?? CH3COO����Na����C2H5OH ? �ڷ�Ӧ�����У������ʵ�Ũ����ʱ����ı䣨ע��Na�������ڷ�Ӧǰ��Ũ�Ȳ��䣩�������������ij�ʼŨ��Ϊa���������Ƶij�ʼŨ��Ϊb����ʱ��Ϊtʱ�����������Ũ�Ⱦ�Ϊx����ʱ�̵ķ�Ӧ�ٶ�Ϊ�� ?ʽ�У�kΪ��Ӧ�����ʳ���������ʽ���ֿɵã� ����ʼŨ��a=b��9��(1)ʽ��Ϊ ��ͬʱ�̸����ʵ�Ũ�ȿ��û�ѧ��������������������Ӧ�е�OH��Ũ�ȣ�Ҳ����������������Һ�ĵ絼����á��ڱ�ʵ�������Dz��ú�һ�ַ��������õ絼�����ⶨ�� �絼�ǵ��嵼�����������ȣ������ĵ������������ɵ����ڵ糡���˶���ʵ�ֵģ����������Һ�ĵ���������������������������Ǩ�ƵĽ�����絼L�ǵ���R�ĵ����� ʽ��AΪ����Ľ������lΪ����ij��ȣ�Lg�Ƶ絼�ʡ��������������ǣ���l=1m��A=1m2ʱ�ĵ絼����һ�ֽ�������һ���¶��£�Lg��һ���ġ��Ե������Һ��Lg�������¶��йأ���������Һ�е�����Ũ���йء����ж������Ӵ��ڵ���Һ�У�Lg�Ǹ�������Ǩ�����õ��ܺͣ�������Һ�����ӵ���Ŀ��������������Լ�����Ǩ�����йء��ڱ�ʵ���У����ڷ�Ӧ���ڽ�ϡ��ˮ��Һ�н��еģ����ǿ��Լٶ�CH3COONaȫ�����룬��Ӧǰ����Һ��������Ŀ������������ɲ��䣬������CH3COO����Ǩ���ʱ�OH����Ǩ����С�����ŷ�Ӧ�Ľ��У�OH�����ϼ��٣�CH3COO����Ũ�Ȳ������ӣ�����ϵ�絼��ֵ����½�����һ����Χ�ڣ�������Ϊ��ϵ�ĵ絼�ʵļ�������CH3COO����Ũ��x�����������ȣ���t=tʱ ʽ��L0Ϊ��ʼʱ�ĵ絼�ʣ�LtΪtʱ�ĵ絼�ʡ���t=t��ʱ��Ӧ����CH3COO����Ũ��Ϊa������ ʽ��L������Ӧ����ʱ�ĵ絼�ʣ�KΪ��������������4������5�����루3��ʽ�ã� ��д�ɣ� �� ��ֱ�߷��̣�6����֪��ֻҪ�ⶨ��L0��L���Լ�һ��Ltֵ������ ��Ӧ�Ļ�ܿɸ��ݰ�������˹��ʽ���㣺 ���ֵã� ʽ��k1��k2�ֱ��Ӧ���¶�T1��T2�ķ�Ӧ���ʳ�����RΪ���峣����EaΪ��Ӧ�Ļ�ܡ� �ġ�������ҩƷ�� ? DDS���͵絼���� 1̨ �絼�� 1ֻ ���²� 1�� 100mL���¼���Ӧ�� 1�� 0.5mL��Һ�� 1֧ 100mL��Һ�� 1֧ 50mL���ձ� 1�� 50mL�ζ��� 1֧ 250mL��ƿ 3�� ��� 1�� ������ 1ֻ CH3COOC2H5�Լ����������� NaOH���������� ��ָ̪ʾ����Һ �塢ʵ�鲽�裺 1�����²�ʹ�������25���0.2�档 2���絼���ǡ����ݸ�¼���絼���ǵ�ʹ�á��Ե絼���ǽ���0�㼰���̶�У���������������õ絼�缫�ij�����������ť���������λ�á� 3��NaOH��Һ�����ƣ��������£� ��һ��С�ձ�����������ŨNaOH��Һ����1000ml�Ĺ��ƿװ��Լ900ml������ˮ������ѡ��ʵ�������IJ����缫����ˮ�У� ��1�����ѡ�õ絼���Dz�������Ž��������£���μ���ŨŨNaOH��Һ��L=1300��1400��S/cm ��2�����ѡ�����ӷ����Dz�������Ž��������£���μ���ŨŨNaOH��Һ��PH=12.00���ҡ� 4��NaOH��Һ�ĵζ����������£� �����ƺõ�NaOH��Һ���˹��ֶ��ζ��ܺͷ�ָ̪ʾ���������½���Ũ�Ȳⶨ���ظ��������ϣ�ȡƽ��ֵ�� 5��L ȡ100ml�����ҵζ��õ�NaOH��Һ���ں��¼���Ӧ���У�����ϴ��������ˮ�IJ����缫������10���ӣ��ȵ絼���ϵĶ����ȶ���ÿ��1���Ӷ�ȡһ�����ݣ��ⶨ����ƽ�е����ݡ� 6��L ���L 7��������Ҫ������������ 8��������5��6��7�ڵڶ����¶��½��в�����������30.00�������35.00�棩 ����ʵ��ע����� 1�� NaOH��Һ�������������ǰӦԤ�Ⱥ��¡� 2�� ��ϴ���缫ʱ��������ֽ���õ缫�ϵIJ��ڡ� �������ݼ�¼�������� 1��NaOH��Һ�ĵζ����ݣ� 2��L 3������ǡ���Ĺ�ϵʽ��ͼ��һֱ�ߣ�������б����Ӧ���ʳ���k�� 4����k25��k30�����ݰ�������˹��ʽ ? �����Ӧ�Ļ��Ea�� �ˡ�˼���⣺ 1.��ѧ��Ӧ����ѧ�о����������棬�ֱ�Ϊ�� �� 2.��ѧ��Ӧ����ѧ��������Ҫ������ʲô��һ���������һ������Ӧ���ȱ�ʵ��ȷ���� �� 3.��Һ���ѧ��Ӧʵ���о�����Ҫ��������ǣ� �� 4.��ѧ��Ӧ�п�������Ϊ����ȷ���������ϱ仯������Ũ�Ȼ�������������ѧ������Ӧ�ù�ע��������Ҫʱ�������: ��. 5.���ڶ���ѧ�о��У��������������������ѡ������Щ����Ŀ��ǣ� �𣺣�1����Ӧǰ����ϵ���������ı仯�dz������� ��2���������Ũ�ȳ����Թ�ϵ������������Ϊ��ѣ� ��3���������Ũ�Ȳ������Թ�ϵ�ķ�����������Ϊ�� 6.���ݾ��м�Ӧ�����Ļ�ѧ��Ӧ����ѧ�������������ļ���ȷ����Ӧ���������ݴ��������� ���ַ������ַ�����˥�ڷ����ı��ʼ����Ũ�ȷ��� 7.����ѧʵ�飬һ�������ΪʲôҪ�ں��������½��У� �𣺲�ͬ�¶��µķ�Ӧ�ٶȲ�һ������Ӧ���ʳ������¶��йأ��絼�ʵ���ֵ���¶�Ҳ�йأ�����Ҫȷ���ж���ѧ�����������ں��������½��С� 8.1999�꣬������A.H.Zewail��ŵ�������Ļɾ��ǣ� ��: ���뼤�⼼���о����컯ѧ��Ӧ���̺���̬�� ���� ����Һ����ѧ��Ӧ����ѧʵ���о� �����������ɲο������еĽ���ժҪ��������ƴ�Ӷ��ɣ����ο�����ϸ�����뿴�й����ϣ� ��ѧ��Ӧ����ѧ�о����������棺 ��1����ѧ��Ӧ�����о�����2����ѧ��Ӧ�����������̣��о��� ��ѧ��Ӧ�����о�����㣺 ���Dz���һ������Ũ����ʱ��ı仯����������Ũ������ȷ��ϵ����������ʱ��ı仯���ߡ� ������Ҫ��ʱ���� ��ѧ��Ӧ�п��������еļ��죬Ҳ�еļ�������Բ�ͬ�����������Ҫʹ�ò�ͬ��ʵ�鷽���Ͳ��������������ѡ��ʵ�鷽���Ͳ�������ʱ����Ҫ���ǵ�����Һ�෴Ӧ����ѧʵ���о���������Ҫע�����Ҫʱ���� 1����ѧ��Ӧ�յ�ʱ��t(i) t(i)��������������� ��1�� �����������ʻ�Ϻ�ѧ��Ӧ���ܽ��У��ӻ�Ͽ�ʼ����ϵ��Ͼ���һ������Ҫ��ʱ�䣻 ��2�� ��ѧ��Ӧ�������������⡢�硢�š��������ߵ��ⳡ��������У���������������ѧ��Ӧ��ʼ����Ҫ��ʱ�䡣 2�����ݲ���ʱ��t(m) t(m)��������������� ��1�� ʵ�������������Ӧʱ�䣺�����Ĵ�����������Ԫ�����ܼ�ת�Ƶ���������Ũ�Ƚϴ����Һ��ʱ�����������ȶ��ɿ���������Ҫ��ʱ�䣻 ��2�� �����ڽ��еĻ�ѧ��Ӧ��ϵ��ȡ��һ���֣������еĻ�ѧ��Ӧֹͣ���Ӳ�����ʼ����ѧ��Ӧ��ȫֹͣ������Ũ�Ȳ�����ʱ��仯������Ҫ��ʱ�䡣 3����ѧ��Ӧʱ�䣨ͨ��ȡ����Ũ�ȵİ�˥��t ���ջ�ѧ����ʽ�����ʺ���������ٵ����������ڷ�Ӧ�����ĵ�һ������Ҫ��ʱ�䡣 Ϊ���ܹ���ʵ���в������ɿ���ȷ�����С�Ķ���ѧ���ݣ���������ʱ��֮������������µ������� t ���t ��1�������ʵ��������� ����Ӧ��t ��2�����ٻ�Ϸ� 1923����Hartridge��Roughton��չ�����ġ� t(i)=0.5��1ms ���������� t ��3����ԥ�����ɳڷ���Ծ�䷨ ��������Manfred ��Eigen ����ͬ�����ڸ�͢���о���չ�����ġ� �ڸ�ʵ�鷽���У��������ʻ��ʱ�䣬�ڷ�Ӧ��ϵ���ﻯѧƽ����̵�ʱ�����ڣ��������⡢�硢�š����ߵȶ������������ı�ԭ��ѧƽ����ϵ��������ʹ��ƫ�뻯ѧƽ�⣬�þ�ƽ��״̬����ƽ��״̬�仯����Ҫ��ʱ������ʾ��ѧ��Ӧ���ʡ� t(i)��1��s ��4�����巨 ���巨���������о��ķ�Ӧ��ϵ�б���ŷ��䣨�⡢�����ߡ�X���ߡ����⣩��������ӣ�ͨ���ǵ��ӣ��ɵ��Ӽ��������������������»������ʣ��缤���ĵ���̬�����ɻ����������ӵȣ���������ѧ��Ӧ�����ڹ⻯ѧ�ͷ��仯ѧ�о���Χ�� ���ʹ��ǿ�Ŀɼ���������߷��䣬ʹ��Ӧ��ϵ���������ɻ�����Ӽ���̬���÷���ͨ����Ϊ������ֽ⡱����1949����Norrish ��Porter �ڽ��Ŵ�ѧ�о������ġ� ��������ƣ� t(i)=10-6s ���ڼ��⣺ t(i)=10-9s �������⼼�����⣺ t(i)=10-12s ���ʹ�õ�������X�������䷴Ӧ��ϵ��ʹ��ϵ�ڵ����ʷ��ӵ��룬����Ϊ��������⡱���������о����ӻ����ʵķ�Ӧ�������Բ����ܼ������ӣ���ˮ�����ӣ� ��5�������ڷ������������� �����ڷ�ʵ�����û�ѧ��ӦӰ�����ߵ���״��Ƶ�ʵķ�������H. Gutowsky����ͬ������1953��ͺ˴Ź�����������ģ����ַ�������ں˴Ź����������ߣ���ʱҲ���ڵ������������ף�������ԭ���Ͽ��������κ����ߣ��ؼ�Ҫ�������㹻�ߵķֱ��ʡ� ���µĻ�ѧ�仯�� ����ת�ƣ�Cu++Cu2+���� Cu2++ Cu+63Cu�˴Ź��� ����ת�ƣ�H2O +OH?���� HO?+ H2O H�˴Ź��� CH3OH + OH?����CH3O?+ H2O H�˴Ź��� ����仯�������� �ţ�����ʽ������ʽ H�˴Ź��� ����������ϵ�ͷ�����t(i)= 0(s)��t(m)��������������������������ͨ���ı���ϵ���¶����ı�t �����ϵ�¶Ƚϸߣ�t �����ϵ�¶����ͣ�t �����ϵ�¶��㹻��ʱ��t ��ͬ����ϵ�õ�������̬����Ƶ�ʷ�ʱ����Ҫ���͵��¶Ȳ�ͬ�����ɱ���Ϊ�仯���ʵIJ�ͬ�� ��6��������µĽ���������������������μ��йط��ӷ�Ӧ��̬ѧ���鼮�� ��ѧ��Ӧ����ϵ��������Ӧ���� ��ѧ��Ӧ����ϵ���뷴Ӧ����֮����ȫ��������ͬ�ĸ����ѧ��Ӧ����ϵ���������ʽṹ�йأ���Ӧ����������ϵ������Ӧ����������Ũ�ȶԷ�Ӧ����Ӱ��̶ȵ�����ָ�ꡣ���ھ��������ۻ�ѧ��Ӧ��˵�������ѧ��Ӧ����ϵ���������뷴Ӧ������������ȣ�Ҳ�Ǽ���żȻ���ɺϣ�û�б�Ȼ����ϵ�� �������۵Ļ�Ԫ��Ӧ����ѧ��Ӧ����ϵ���뵥��Ӧ�ķ�Ӧ����֮�䱻��Ϊ�ж�Ӧ��ϵ�� ��Ԫ��Ӧ�����������ö��ɡ����ۿ�����ԭ�� ��Ԫ��Ӧ A+B �������ö��� r+=k+CACB r��=k��CCCD �ۿ�����ԭ�� r= r+?r��= k+CACB?k��CCCD ֻ�л�ѧ��Ӧ�տ�ʼ���õ�һС��ʱ���ڣ�����C��D��Ũ�Ⱥ�С������Ӧ�����ʼ������Ժ��Բ���ʱ r(t��0)�� r+�� k+CACB �ɴ˿ɼ������м�Ӧ�����Ļ�ѧ��Ӧ����ѧ���������ʹ�ǻ�Ԫ��Ӧ��Ӧ��Ҳ�ǽ��ƵĽ������ ���м�Ӧ�����Ļ�ѧ��Ӧ�Ķ���ѧ���� �μ���������ѧ�����²ᣩ���ײʵȣ��ߵȽ��������磬P700��734 ����Һ����ද��ѧʵ���о�������Ҫ���� 1�� ��������ʹ�õĻ�ѧ��Һ����������Ҫ�ⶨ��ȷŨ�ȣ� 2�� ���е���ϵ��Ҫ������ѧ��Ӧ��ʼǰij����Һ����ʼ������Z(t=0)�� 3�� ������ѧ��Ӧ��ʼ������Ũ��C��ʱ��t�ı仯����������Ũ���йص������� Z��C����ʱ��t�ı仯���� 4�� ���е���ϵ��Ҫ������ѧ��Ӧ����ƽ���Ժ����Һ��������Z(t=��)�� 5�� ���ݾ��м�Ӧ�����Ļ�ѧ��Ӧ�Ķ���ѧ��������2��3��4���������Ľ�������Ӧ�Ĺ�ʽ�̣�ȷ����Ӧ����n�� 6�� ����ijȷ���¶��µĻ�ѧ��Ӧ���ʳ���k�� 7�� �仯��ͬ���¶��������вⶨ����k��T�Ĺ�ϵ���㻯ѧ��Ӧ���Ea�� �뻯ѧ��Ӧ����ѧ�����йص�ŵ������ �ο����ϣ� 1����������ѧ�����²ᣩ���ײʵȣ��ߵȽ��������� 2����������ѧ��(��)V.����µ��� �ߵȽ��������磬 1983��7�� 3����չ��21���͵Ļ�ѧ�������ɵ����࣬��ѧ��ҵ�����磬20##��5��
�ڶ�ƪ��10.��������������Ӧ����ѧ�о�
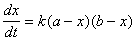 (10��1)
(10��1) (10��2)
(10��2)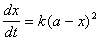 �����ֵã�
�����ֵã�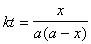 (10��3)
(10��3)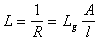
 (10��4)
(10��4)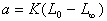 (10��5)
(10��5)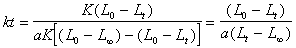
 (10��6)
(10��6)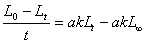 (10��7)
(10��7)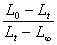 ��t��ͼ��Ӧ��һֱ�ߣ�ֱ�ߵ�б�ʾ��Ƿ�Ӧ�ٶȺͳ�ʼŨ��a�ij˻���k�ĵ�λΪdm3��mol-1��min-1��
��t��ͼ��Ӧ��һֱ�ߣ�ֱ�ߵ�б�ʾ��Ƿ�Ӧ�ٶȺͳ�ʼŨ��a�ij˻���k�ĵ�λΪdm3��mol-1��min-1��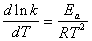 (10��8)
(10��8)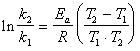 (10��9)
(10��9) ����PH
����PH ���IJⶨ��������25.00�������30.00�棩
���IJⶨ��������25.00�������30.00�棩 ����PH
����PH ���IJⶨ��������25.00�������30.00�棩
���IJⶨ��������25.00�������30.00�棩 ����PH
����PH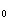 ���IJⶨ��ʹ��С��������Һ����ȡ��������������������������ڲ����ף�����������ȫ��������Һ�У���Ҫ�����ڲ������ڱ��ϣ�����Ũ�Ȳ����ŵ�һ��ʱ�������ʱ������ƽ�ȱ仯���������һ�����ݣ��Ժ�ÿ��1���Ӷ�һ������15���Ӻ�ÿ��2���Ӷ�һ���������е�35���Ӻ������
���IJⶨ��ʹ��С��������Һ����ȡ��������������������������ڲ����ף�����������ȫ��������Һ�У���Ҫ�����ڲ������ڱ��ϣ�����Ũ�Ȳ����ŵ�һ��ʱ�������ʱ������ƽ�ȱ仯���������һ�����ݣ��Ժ�ÿ��1���Ӷ�һ������15���Ӻ�ÿ��2���Ӷ�һ���������е�35���Ӻ������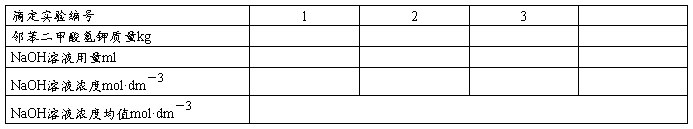
 ����PH
����PH ����L
����L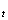 ����PH
����PH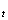 ���IJⶨ�� ʵ���¶ȣ���
���IJⶨ�� ʵ���¶ȣ���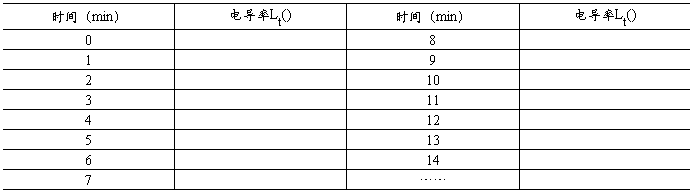
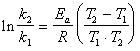
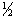 ��
��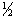 ��t(i) �� t
��t(i) �� t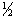 ��t(m)
��t(m) ��С���ͱ�������Ӧ�����ʻ�Ϸ������ԥ����ͬʱ�и�ʱ��ֱ��ʵ�ʵ�������������ʹ���������ڲ�ͬ��ѧ��Ӧ���ʲⶨ�Ķ���ʵ�鷽����
��С���ͱ�������Ӧ�����ʻ�Ϸ������ԥ����ͬʱ�и�ʱ��ֱ��ʵ�ʵ�������������ʹ���������ڲ�ͬ��ѧ��Ӧ���ʲⶨ�Ķ���ʵ�鷽���� �������ӣ������ϣ���ͨ����������
�������ӣ������ϣ���ͨ���������� =101��103ms��
=101��103ms�� ��
�� ��t(m)���������ڲ���ʱ���ڸ��ٿ��ٱ仯������״̬����ȷ����̬������Ƶ�ʷ壬��t(m)ʱ����õ�����������̬��ƽ���������ֻ��һ�������ˣ�
��t(m)���������ڲ���ʱ���ڸ��ٿ��ٱ仯������״̬����ȷ����̬������Ƶ�ʷ壬��t(m)ʱ����õ�����������̬��ƽ���������ֻ��һ�������ˣ� ���������̬���ȶ�����ʱ��䳤�������ͻ�Ӳ��ֱܷ�������̬��Ϊ���Էֱ�������̬��
���������̬���ȶ�����ʱ��䳤�������ͻ�Ӳ��ֱܷ�������̬��Ϊ���Էֱ�������̬�� ��t(m)�������ܹ��ڲ���ʱ���ڷֱ���������״̬����ȷ����̬������Ƶ�ʷ壬�ͻ�õ��������շ��ˡ�
��t(m)�������ܹ��ڲ���ʱ���ڷֱ���������״̬����ȷ����̬������Ƶ�ʷ壬�ͻ�õ��������շ��ˡ� C+D
C+D